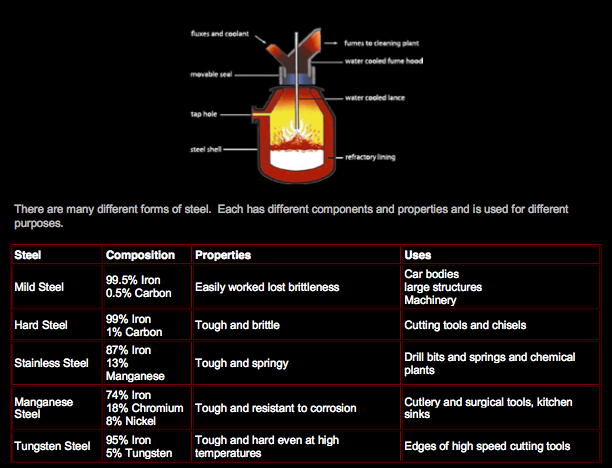
Conversion of Iron into Steel:
Iron produced in the blast furnace is called pig iron. It contains 4% carbon as well as other impurities such as sulfur, silicon and phosphorus which make it hard and brittle. It got that name from the fact that it has to be poured into mould called pigs before it is converted into steel. Most of produced iron is converted into steel because steel has better properties.
Making steel out of pig iron is a process done in a basic oxygen furnace:
Iron produced in the blast furnace is called pig iron. It contains 4% carbon as well as other impurities such as sulfur, silicon and phosphorus which make it hard and brittle. It got that name from the fact that it has to be poured into mould called pigs before it is converted into steel. Most of produced iron is converted into steel because steel has better properties.
Making steel out of pig iron is a process done in a basic oxygen furnace:
- Molten pig iron is poured into the oxygen furnace
- A water cooled lance is introduced which blows oxygen onto the surface of the molten iron
- Impurities start to react
- Carbon is oxidized into carbon monoxide and carbon dioxide and escape
- Sulfur is oxidized into sulfur dioxide and escapes
- Silicon and phosphorus are oxidized into silicon oxide and phosphorus pentoxide which are solids.
- Calcium oxide (lime) is added to remove the solid impurities as slag which is skimmed off the surface
- Throughout the process, sample of the iron are being taken and analyzed for the percentage of carbon
present in it. When the percentage of carbon desired is reached, the furnace is switched off and the steel is collected.
Extraction of Zinc:
The ore of zinc is called zinc blende and it is made of zinc sulfide. Zinc is obtained from zinc sulfide by converting it into zinc oxide then reducing it using coke, but first zinc sulfide must be concentrated.
Zinc sulfide from zinc blende is concentrated by a process called froth floatation. In this process, the ore is crushed
and put into tanks of water containing a frothing agent which makes the mixture froth up. Hot air is blown in and froth starts to form. Rock impurities in the ore get soaked and sink to the bottom of the tank. Zinc sulfide particles cannot be soaked by water; they are lifted by the bubbles of air up with the froth and are then skimmed off. This is now concentrated zinc sulfide.
Then, zinc sulfide gets heated very strongly with hot air in a furnace. Zinc sulfide reacts with oxygen from the air to produce zinc oxide and sulfur dioxide gas which escapes as waste gas.
2ZnS + 3O2 → 2ZnO + 2SO2 Sulfur dioxide is used in the manufacture of sulfuric acid.
Zinc oxide produced is put into a furnace with powdered coke. The mixture is heated till 1400oC. Carbon from the coke reduces the zinc oxide into zinc producing carbon monoxide which escapes as waste gas.
ZnO + C → Zn + CO
The ore of zinc is called zinc blende and it is made of zinc sulfide. Zinc is obtained from zinc sulfide by converting it into zinc oxide then reducing it using coke, but first zinc sulfide must be concentrated.
Zinc sulfide from zinc blende is concentrated by a process called froth floatation. In this process, the ore is crushed
and put into tanks of water containing a frothing agent which makes the mixture froth up. Hot air is blown in and froth starts to form. Rock impurities in the ore get soaked and sink to the bottom of the tank. Zinc sulfide particles cannot be soaked by water; they are lifted by the bubbles of air up with the froth and are then skimmed off. This is now concentrated zinc sulfide.
Then, zinc sulfide gets heated very strongly with hot air in a furnace. Zinc sulfide reacts with oxygen from the air to produce zinc oxide and sulfur dioxide gas which escapes as waste gas.
2ZnS + 3O2 → 2ZnO + 2SO2 Sulfur dioxide is used in the manufacture of sulfuric acid.
Zinc oxide produced is put into a furnace with powdered coke. The mixture is heated till 1400oC. Carbon from the coke reduces the zinc oxide into zinc producing carbon monoxide which escapes as waste gas.
ZnO + C → Zn + CO
Carbon monoxide produced is hot and is used to heat the furnace to reduce heating costs. The pure zinc produced is collected and left to cool down. Zinc is used in many ways like the production of the alloy brass, galvanization and making car batteries.
Uses of Zinc:
Making brass
Galvanizing
Extraction of Copper:
Copper is one of the most popular metals. Native copper occurs in some regions in the world. Otherwise, copper exists in its ore, copper pyrites (2CuFeS2). You have studied before that copper can be purified by electrolysis. It can also be extracted from it ore by converting pyrites into copper sulfide by reacting it with oxygen:
2CuFeS2 + 4O2 → Cu2S + 3SO2 + 2FeO
Sulfur oxide produced escapes as waste gas and iron oxide impurities are removed by heating the mixture with silicon converting it in to iron silicate which is run off. The remaining copper sulfide is then heated strongly with air.
Copper sulfide reacts with oxygen from air producing sulfur oxide which escapes as waste gas and pure copper.
Thus copper is extracted.
Uses of Copper:
Cu2S+O2 →2Cu+SO2
Uses of Zinc:
Making brass
Galvanizing
Extraction of Copper:
Copper is one of the most popular metals. Native copper occurs in some regions in the world. Otherwise, copper exists in its ore, copper pyrites (2CuFeS2). You have studied before that copper can be purified by electrolysis. It can also be extracted from it ore by converting pyrites into copper sulfide by reacting it with oxygen:
2CuFeS2 + 4O2 → Cu2S + 3SO2 + 2FeO
Sulfur oxide produced escapes as waste gas and iron oxide impurities are removed by heating the mixture with silicon converting it in to iron silicate which is run off. The remaining copper sulfide is then heated strongly with air.
Copper sulfide reacts with oxygen from air producing sulfur oxide which escapes as waste gas and pure copper.
Thus copper is extracted.
Uses of Copper:
Cu2S+O2 →2Cu+SO2
- In electrical wires because it is a perfect electrical conductor and very ductile, malleable and cheap
- Making alloys such as bronze and brass
- Cooking utensils because it conducts heat and it is has high melting and boiling points and also resists
corrosion
- Electrodes because it is a good conductor of electricity
- Water pipes because it is resistant to corrosion