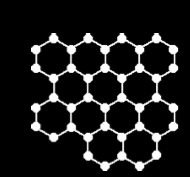
- the giant covalent structures of graphite and diamond
Diamond:
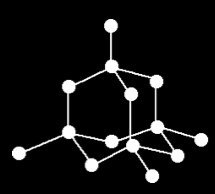
In diamond’s structure, each carbon atom is covalently bonded to four other carbon atoms by very strong bonds forming a 3D tetrahedral shape.
The physical properties of diamond:
which could occur between solvent molecules and carbon atoms which could outweigh the attractions between the covalently bound carbon atoms.
The physical properties of diamond:
- Has a very high melting point (almost 4000°C). Very strong carbon- carbon covalent bonds have to be broken throughout the structure before melting occurs,
- Is very hard. This is again due to the need to break very strong covalent bonds operating in 3-dimensions,
- Doesn't conduct electricity. All the electrons are held tightly between the atoms, and aren't free to move,
- Is insoluble in water and organic solvents. There are no possible attractions
which could occur between solvent molecules and carbon atoms which could outweigh the attractions between the covalently bound carbon atoms.
Graphite:
In the graphite structure, each carbon atom is strongly bonded covalently to three other carbon atoms forming layers of linked hexagons. Each layer acts as a molecule, the intermolecular forces between the layers is very weak allowing layers to slide over each other. This makes graphite a good lubricant.
The physical properties of graphite:
Has a high melting point, similar to that of diamond. In order to melt graphite, it isn't enough to loosen one sheet from another.
You have to break the covalent bonding throughout the whole structure,
Has a soft, slippery feel, and is used in pencils and as a dry lubricant for things like locks. You can think of graphite
rather like a pack of cards - each card is strong, but the cards will slide over each other, or even fall off the pack
altogether. When you use a pencil, sheets are rubbed off and stick to the paper,
In the graphite structure, each carbon atom is strongly bonded covalently to three other carbon atoms forming layers of linked hexagons. Each layer acts as a molecule, the intermolecular forces between the layers is very weak allowing layers to slide over each other. This makes graphite a good lubricant.
The physical properties of graphite:
Has a high melting point, similar to that of diamond. In order to melt graphite, it isn't enough to loosen one sheet from another.
You have to break the covalent bonding throughout the whole structure,
Has a soft, slippery feel, and is used in pencils and as a dry lubricant for things like locks. You can think of graphite
rather like a pack of cards - each card is strong, but the cards will slide over each other, or even fall off the pack
altogether. When you use a pencil, sheets are rubbed off and stick to the paper,
- Has a lower density than diamond. This is because of the relatively large amount of space that is "wasted"
between the sheets,
- Is insoluble in water and organic solvents - for the same reason that diamond is insoluble. Attractions between
solvent molecules and carbon atoms will never be strong enough to overcome the strong covalent bonds in
graphite,
- Conducts electricity. The delocalised electrons are free to move throughout the sheets. If a piece of graphite is
connected into a circuit, electrons can fall off one end of the sheet and be replaced with new ones at the other end.
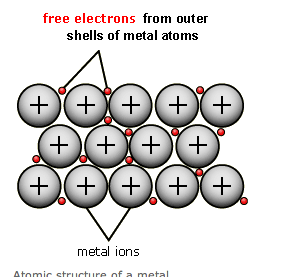
metallic bonding - higher
Giant structures with free electronsMetals form giant structures in which electrons in the outer shells of the metalatoms are free to move. The metallic bond is the force of attraction between these free electrons and metal ions. Metallic bonds are strong, so metals can maintain a regular structure and usually have high melting and boiling points.
Metals are good conductors of electricity and heat, because the free electrons carry a charge or heat energy through the metal. The free electrons allow metal atoms to slide over each other, so metals are malleable and ductile.