Stoichiometry
Stoichiometry deals with the relative quantities of reactants and products in chemical reactions. In a balanced chemical reaction, the relations among quantities of reactants and products typically form a ratio of whole numbers. For example, in a reaction that forms ammonia (NH3), exactly one molecule of nitrogen (N2) reacts with three molecules of hydrogen (H2) to produce two molecules of NH3
N2 + 3H2 → 2NH3
Stoichiometry can be used to calculate quantities such as the amount of products (in mass, moles, volume, etc.) that can be produced with given reactants and percent yield (the percentage of the given reactant that is made into the product). Stoichiometry calculations can predict how elements and components diluted in a standard solution react in experimental conditions. Stoichiometry is founded on the law of conservation of mass: the mass of the reactants
equals the mass of the products.
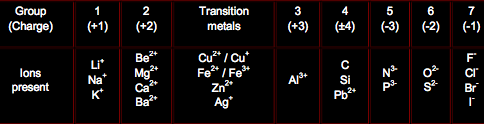
The Periodic Table and Charges:
N2 + 3H2 → 2NH3
Stoichiometry can be used to calculate quantities such as the amount of products (in mass, moles, volume, etc.) that can be produced with given reactants and percent yield (the percentage of the given reactant that is made into the product). Stoichiometry calculations can predict how elements and components diluted in a standard solution react in experimental conditions. Stoichiometry is founded on the law of conservation of mass: the mass of the reactants
equals the mass of the products.
The Periodic Table and Charges:
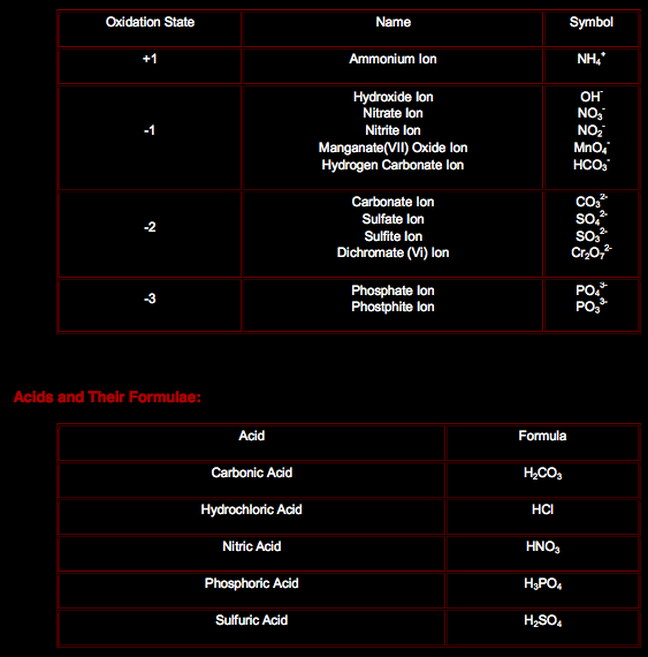
Compound Ions:
They are like molecules but with a charge on them:
Chemical Changes:
When chemical reactions take place, a certain change is certain. This change could be in color, energy or something like bubbles or a precipitate formation.
Color Change in Reactions:
When iron, which is grey, reacts with sulfur, which is yellow, a black solid, iron sulfide is formed. The color changed
from grey and yellow to black.
When chemical reactions take place, a certain change is certain. This change could be in color, energy or something like bubbles or a precipitate formation.
Color Change in Reactions:
When iron, which is grey, reacts with sulfur, which is yellow, a black solid, iron sulfide is formed. The color changed
from grey and yellow to black.
Iron + Sulphur → I ron Sulfide
(grey fillings) (yellow powder) (black solid)
(grey fillings) (yellow powder) (black solid)
Energy Change in Reactions:
Energy is not necessarily heat, there are several forms of energy like Light and sound energy too.
When magnesium reacts with oxygen in air to make a combustion reaction, it burns with a very bright flame forming magnesium oxide. This bright flame is light energy which indicates a reaction took place.
Some reactions also release energy in form of sound like explosive reactions.
Effervescence in Reactions:
In some reactions, bubbles are formed rapidly. Those bubbles are indication that a gas was formed which is a new substance, this means a reaction took place.
When calcium carbonate reacts with hydrochloric acid, carbon dioxide gas is released in bubbles.
Energy is not necessarily heat, there are several forms of energy like Light and sound energy too.
When magnesium reacts with oxygen in air to make a combustion reaction, it burns with a very bright flame forming magnesium oxide. This bright flame is light energy which indicates a reaction took place.
Some reactions also release energy in form of sound like explosive reactions.
Effervescence in Reactions:
In some reactions, bubbles are formed rapidly. Those bubbles are indication that a gas was formed which is a new substance, this means a reaction took place.
When calcium carbonate reacts with hydrochloric acid, carbon dioxide gas is released in bubbles.
CaCO3 + 2HCl → CaCl2 + H2O + CO2
Precipitation in Reactions:
When two solutions react together, sometimes one of the products is an insoluble solid called precipitate. When silver nitrate solution reacts with a potassium chloride solution, white insoluble silver chloride is formed which is a precipitate.
AgNO3 + KCl → AgCl + KNO3
Writing and Balancing Equations:
Chemical reactions are always represented by chemical equations to show the reactants and the products. There are two types of chemical equations. These are word equations and symbolic equations.
For example if we want to represent the reaction between sodium and chlorine which produces sodium chloride as a
word equation it will be like this:
Sodium + Chlorine → Sodium Chloride
If we want to represent the same reaction by a symbolic equation it will be like this:
Na + Cl2 → NaCl
The above symbolic equation however, is not chemically accurate. This is why chemists prefer to use what’s called a balanced symbolic equation. In this type of equation, we make sure that the number of atoms of each element in the reactants is the same in the products. If you look at the equation above you will find that we have one Na atom in the reactants and one Na+ ion in the product, but we have 2 Cl atoms in the reactants and only one Cl ion in the
product. This is why this equation needs to be balanced as follows:
2Na + Cl2 → 2NaCl
When two solutions react together, sometimes one of the products is an insoluble solid called precipitate. When silver nitrate solution reacts with a potassium chloride solution, white insoluble silver chloride is formed which is a precipitate.
AgNO3 + KCl → AgCl + KNO3
Writing and Balancing Equations:
Chemical reactions are always represented by chemical equations to show the reactants and the products. There are two types of chemical equations. These are word equations and symbolic equations.
For example if we want to represent the reaction between sodium and chlorine which produces sodium chloride as a
word equation it will be like this:
Sodium + Chlorine → Sodium Chloride
If we want to represent the same reaction by a symbolic equation it will be like this:
Na + Cl2 → NaCl
The above symbolic equation however, is not chemically accurate. This is why chemists prefer to use what’s called a balanced symbolic equation. In this type of equation, we make sure that the number of atoms of each element in the reactants is the same in the products. If you look at the equation above you will find that we have one Na atom in the reactants and one Na+ ion in the product, but we have 2 Cl atoms in the reactants and only one Cl ion in the
product. This is why this equation needs to be balanced as follows:
2Na + Cl2 → 2NaCl
The Mole Concept:
A mole is a unit to count the number of atoms, ions or molecules. They believed that, for example, if one molecule of carbon dioxide (CO2) contained 1 carbon atom and 2 oxygen atoms, then the ratio of carbon atoms to oxygen atoms is 1:2. So
A mole is a unit to count the number of atoms, ions or molecules. They believed that, for example, if one molecule of carbon dioxide (CO2) contained 1 carbon atom and 2 oxygen atoms, then the ratio of carbon atoms to oxygen atoms is 1:2. So
Chemists use a method similar to that one, but on a larger scale, in industries to prevent wasting money by buying excess substances that will not be used. This is called Avogadro’s Constant.
Avogadro’s Constant in Solids:
Avogadro was a scientist in the 19th century. He discovered a relationship between a certain amount of substance (atoms, ions or molecules) and the Ar (Relative atomic mass) or Mr (Relative Molecular Mass) of the substance.
Avogadro was a scientist in the 19th century. He discovered a relationship between a certain amount of substance (atoms, ions or molecules) and the Ar (Relative atomic mass) or Mr (Relative Molecular Mass) of the substance.
The Mr of a compound is the sum of the Ar of all the atoms present in one molecule of the compound.
What Avogadro discovered is that if I am holding 6x1023 atoms in my hand, its mass is equal to the Ar of Iron (Fe). This unit is called Mole.
From this we conclude that the mass of one mole of any substance is the Ar of it (if it was an element) or the Mr of it (if it was a compound). The mass of one mole of any substance is expressed as the molar mass, and the word mole can be abbreviated with mol. The molar mass is always expressed in grams.
Chemical change
In a chemical change, a new chemical substance is produced.
The difference between a mixture and a compound
Mixture: 2 substances are mixed together but not chemically bonded.
Compound: 2 substances are chemically bonded together
The signs of a chemical change
A chemical change is usually called a chemical reaction. You can tell when a chemical reaction has taken place by these signs:
1. Once or more new chemical substances are formed
The new substance usually looks different from the starting substances.
2. Energy is taken in or given out during the reaction.
A change that gives out heat energy is called exothermic A change that takes in heat energy is called endothermic
3. The change is usually difficult to reverse.
This means it will be hard to get back the raw materials of the reaction.
The difference between a mixture and a compound
Mixture: 2 substances are mixed together but not chemically bonded.
Compound: 2 substances are chemically bonded together
The signs of a chemical change
A chemical change is usually called a chemical reaction. You can tell when a chemical reaction has taken place by these signs:
1. Once or more new chemical substances are formed
The new substance usually looks different from the starting substances.
2. Energy is taken in or given out during the reaction.
A change that gives out heat energy is called exothermic A change that takes in heat energy is called endothermic
3. The change is usually difficult to reverse.
This means it will be hard to get back the raw materials of the reaction.
Physical change
If no new chemical substance is formed, a change is a physical change.
Equations for chemical reactions
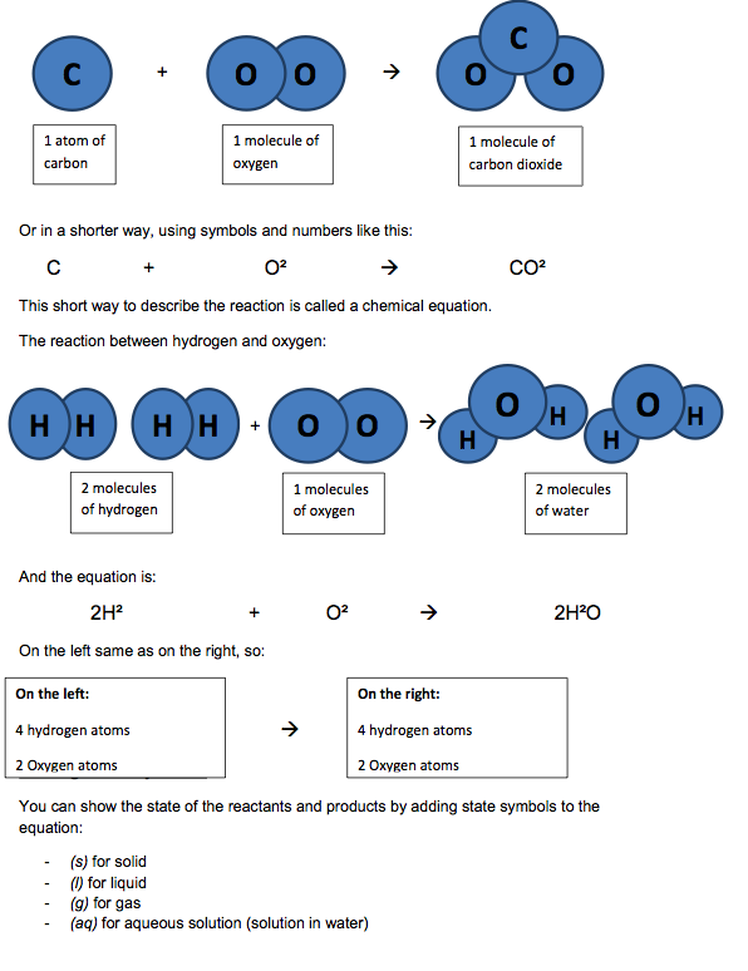
The reaction between carbon and oxygen. When they react together, they form carbon dioxide. Carbon and oxygen are the reactants. Carbon dioxide is the product of the reaction.
Equations for chemical reactions
The reaction between carbon and oxygen. When they react together, they form carbon dioxide. Carbon and oxygen are the reactants. Carbon dioxide is the product of the reaction.