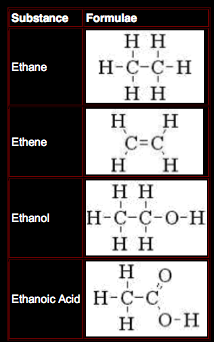
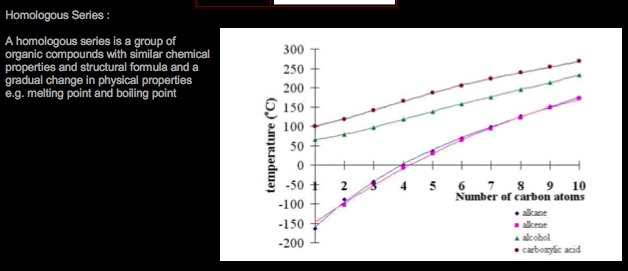
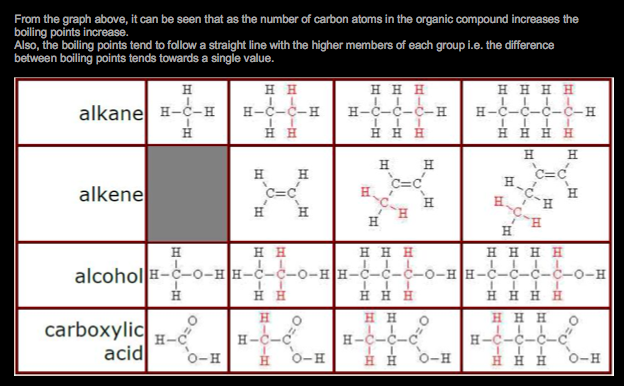
Organic Chemistry
Structural formulae :
As well as using a normal type of molecular formula to describe an organic molecule, they can be represented by drawing out their structure i.e. by showing how the atoms are connected, or bonded, to each other.
In order to do this a few rules have to be followed –
o carbon atoms must be bonded four times;
o oxygen atoms must be bonded twice;
o hydrogen atoms must bond only once.
As well as using a normal type of molecular formula to describe an organic molecule, they can be represented by drawing out their structure i.e. by showing how the atoms are connected, or bonded, to each other.
In order to do this a few rules have to be followed –
o carbon atoms must be bonded four times;
o oxygen atoms must be bonded twice;
o hydrogen atoms must bond only once.
Alkane:
Alkanes are the simplest homologous series of compounds and their names follow this pattern, CH4 - methane
C2H6 - ethane
C3H8 - propane
C4H10 - butane C5H12 – pentane
The general chemical formula for an alkane is CnH2n+2.
Branched alkanes -
When the alkane is not just a simple straight chain of carbon atoms joined together the names become a little more complex.
The longest connected chain of carbon atoms must be found as before and the alkane name generated as usual. Then the name for the pendent group is found, again by counting the number of carbon atoms present, and used as a prefix.
CH3- group : methyl___
CH3CH2- group : ethyl___
CH3CH2CH2- group : propyl___
CH3CH2CH2CH2- group : butyl___
Reactions of alkanes :
(i) Combustion -
Alkanes, along with all other types of hydrocarbon, will burn in an excess of oxygen to give carbon dioxide and water only as the products,
e.g. CH4(g) + 2O2(g) CO2(g) + 2H2O(g)
in general,
CnH2n+2(g) + (1.5n+0.5)O2(g) nCO2(g) + (n+1)H2O(g)
If there is not enough oxygen present then instead of carbon dioxide, carbon monoxide, CO, is produced. Carbon monoxide is particularly toxic and absorbed into blood, through respiration, very easily. For domestic heating systems
it is particularly important that enough air can get to the flame to avoid carbon monoxide being generated in the home. Car engines also require a lot of air and there is a lot of research going on to make the internal combustion engine more efficient, and so put out less carbon monoxide.
Note also that both alkanes and carbon dioxide are green house gases, i.e. they trap infra-red (i.-r.) radiation inside the Earth's atmosphere, gradually increasing global temperatures.
(ii) Halogenation -
The only other reaction that an alkane will undergo is a reaction with a halogen (chlorine or bromine typically ) with UV light present as an initiator of the reaction,
e.g. CH4(g) + Br2(g) CH3Br(g) + HBr(g)
The UV light causes the formation of free radical halogen atoms by providing enough energy for the bond between the two halogen atoms to break. A halogen atom attacks the alkane, substituting itself for a hydrogen atom. This substitution may occur many times in an alkane before the reaction is finished.
A similar process occurs high up in the earth's atmosphere when CFC's and other organic solvents react with intense sunlight to produce free radicals, chlorine atoms in this case. These attack molecules of ozone ( O3 ) depleting ozone's concentration and leading to the "holes".
Alkanes are the simplest homologous series of compounds and their names follow this pattern, CH4 - methane
C2H6 - ethane
C3H8 - propane
C4H10 - butane C5H12 – pentane
The general chemical formula for an alkane is CnH2n+2.
Branched alkanes -
When the alkane is not just a simple straight chain of carbon atoms joined together the names become a little more complex.
The longest connected chain of carbon atoms must be found as before and the alkane name generated as usual. Then the name for the pendent group is found, again by counting the number of carbon atoms present, and used as a prefix.
CH3- group : methyl___
CH3CH2- group : ethyl___
CH3CH2CH2- group : propyl___
CH3CH2CH2CH2- group : butyl___
Reactions of alkanes :
(i) Combustion -
Alkanes, along with all other types of hydrocarbon, will burn in an excess of oxygen to give carbon dioxide and water only as the products,
e.g. CH4(g) + 2O2(g) CO2(g) + 2H2O(g)
in general,
CnH2n+2(g) + (1.5n+0.5)O2(g) nCO2(g) + (n+1)H2O(g)
If there is not enough oxygen present then instead of carbon dioxide, carbon monoxide, CO, is produced. Carbon monoxide is particularly toxic and absorbed into blood, through respiration, very easily. For domestic heating systems
it is particularly important that enough air can get to the flame to avoid carbon monoxide being generated in the home. Car engines also require a lot of air and there is a lot of research going on to make the internal combustion engine more efficient, and so put out less carbon monoxide.
Note also that both alkanes and carbon dioxide are green house gases, i.e. they trap infra-red (i.-r.) radiation inside the Earth's atmosphere, gradually increasing global temperatures.
(ii) Halogenation -
The only other reaction that an alkane will undergo is a reaction with a halogen (chlorine or bromine typically ) with UV light present as an initiator of the reaction,
e.g. CH4(g) + Br2(g) CH3Br(g) + HBr(g)
The UV light causes the formation of free radical halogen atoms by providing enough energy for the bond between the two halogen atoms to break. A halogen atom attacks the alkane, substituting itself for a hydrogen atom. This substitution may occur many times in an alkane before the reaction is finished.
A similar process occurs high up in the earth's atmosphere when CFC's and other organic solvents react with intense sunlight to produce free radicals, chlorine atoms in this case. These attack molecules of ozone ( O3 ) depleting ozone's concentration and leading to the "holes".
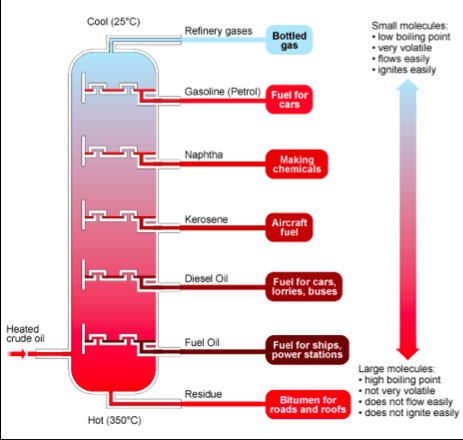
Fractional Distillation:
Crude oil is a mixture of many different hydrocarbon compounds, some of them liquid and some of them gases. These compounds can be separated because the different length of alkanes will have different boiling points. The crude oil is heated up to about 350 C and is fed into a fractionating column, as in the diagram below
Crude oil is a mixture of many different hydrocarbon compounds, some of them liquid and some of them gases. These compounds can be separated because the different length of alkanes will have different boiling points. The crude oil is heated up to about 350 C and is fed into a fractionating column, as in the diagram below
The vapours with the lowest boiling points pass all the way up the column and come off as gases, e.g. methane, ethane and propane. The temperature of the column gradually decreases the higher up the vapours go, and so various fractions will condense to liquids at different heights.
The fractions with the highest boiling points do not vaporize and are collected at the bottom of the fractionating column, e.g. bitumen
Cracking
In industry the fractions obtained from the fractional distillation of crude oil are heated at high pressure in the presence of a catalyst to produce shorter chain alkanes and alkenes. e.g. C10H22 C5H12 + C5H10
Alkane
Alkenes all have a C=C double bond in their structure and their names follow this pattern,
C2H4 – ethane C3H6 – propene C4H8 – butane C5H10 - pentene
The general chemical formula for an alkene is CnH2n Addition reactions of alkenes :
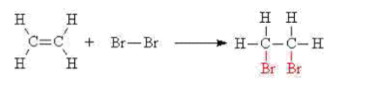
The double bond of an alkene will undergo an addition reaction with aqueous bromine to give a dibromo compound. The orange bromine water is decolourised in the process.
e.g. ethene reacts with bromine water to give 1,2-dibromoethane,
The fractions with the highest boiling points do not vaporize and are collected at the bottom of the fractionating column, e.g. bitumen
Cracking
In industry the fractions obtained from the fractional distillation of crude oil are heated at high pressure in the presence of a catalyst to produce shorter chain alkanes and alkenes. e.g. C10H22 C5H12 + C5H10
Alkane
Alkenes all have a C=C double bond in their structure and their names follow this pattern,
C2H4 – ethane C3H6 – propene C4H8 – butane C5H10 - pentene
The general chemical formula for an alkene is CnH2n Addition reactions of alkenes :
The double bond of an alkene will undergo an addition reaction with aqueous bromine to give a dibromo compound. The orange bromine water is decolourised in the process.
e.g. ethene reacts with bromine water to give 1,2-dibromoethane,
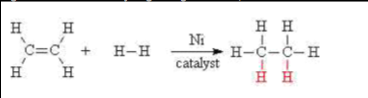
Hydrogenation -
Alkenes may be turned into alkanes by reacting the alkene with hydrogen gas at a high temperature and high pressure. A nickel catalyst is also needed to accomplish this addition reaction.
e.g. ethene reacts with hydrogen to give ethane,
Alkenes may be turned into alkanes by reacting the alkene with hydrogen gas at a high temperature and high pressure. A nickel catalyst is also needed to accomplish this addition reaction.
e.g. ethene reacts with hydrogen to give ethane,
This reaction is also called saturation of the double bond. In ethene the carbon atoms are said to be unsaturated. In ethane the carbon atoms have the maximum number of hydrogen atoms bonded to them, and are said to be saturated.
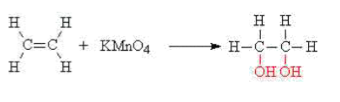
Oxidation -
The carbon-carbon double bond may also be oxidised i.e. have oxygen added to it.
This is accomplished by using acidified potassium manganate(VII) solution at room temperature and pressure. The purple manganate(VII) solution is decolourised during the reaction.
e.g. ethene reacts with acidified potassium manganate(VII)(aq) to give ethan-1,2-diol,
The carbon-carbon double bond may also be oxidised i.e. have oxygen added to it.
This is accomplished by using acidified potassium manganate(VII) solution at room temperature and pressure. The purple manganate(VII) solution is decolourised during the reaction.
e.g. ethene reacts with acidified potassium manganate(VII)(aq) to give ethan-1,2-diol,
Addition Polymerisation
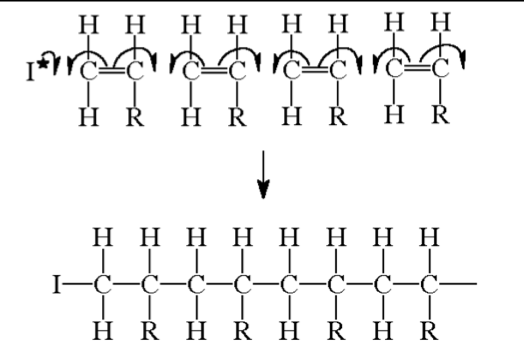
All alkenes will react with free radical initiators to form polymers by a free radicaladdition reaction. Some definitions - monomer - a single unit e.g. an alkene.
All alkenes will react with free radical initiators to form polymers by a free radicaladdition reaction. Some definitions - monomer - a single unit e.g. an alkene.
The reaction progresses by the separate units joining up to form giant, long chains
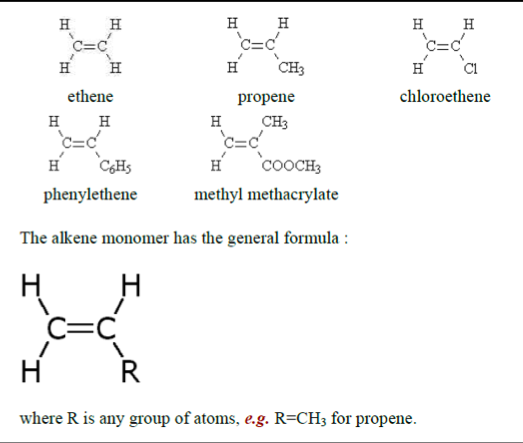
polymer- a material produced from many separate single monomer units joined up together. An addition polymer is simply named after the monomer alkene that it is prepared from,
e.g. ethene makes poly(ethene)
propene makes poly(propene)
phenylethene makes poly(phenylethene) chloroethene makes poly(chloroethene) methyl acrylate makes poly(methyl acrylate)
The structure above shows just 4 separate monomer units joined together. In a real polymer, however, there could be 1000's of units joined up to form the chains. This would be extremely difficult to draw out and so the structure is often shortened to a repeat unit. There are 3 stages to think about when drawing a repeat unit for a polymer –
e.g. ethene makes poly(ethene)
propene makes poly(propene)
phenylethene makes poly(phenylethene) chloroethene makes poly(chloroethene) methyl acrylate makes poly(methyl acrylate)
The structure above shows just 4 separate monomer units joined together. In a real polymer, however, there could be 1000's of units joined up to form the chains. This would be extremely difficult to draw out and so the structure is often shortened to a repeat unit. There are 3 stages to think about when drawing a repeat unit for a polymer –
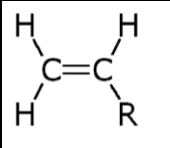
Draw the structure of the desired monomer :
Change the double bond into a single bond and draw bonds going left and right from the carbon atoms :
Place large brackets around the structure and a subscript n and there is the repeat unit.
where R= H for ethene
= CH3 for propene
= C6H5 for phenylethene
= Cl for chloroethene
= COOCH3 for methyl acrylate
Polymer structure and properties –
When the individual alkene units join together to give a polymer they result in the formation of long chains of carbon atoms joined together. In any sample of a polymer there are many separate chains present. These chains will be of varying lengths, depending on the number of alkene units that make them up. These separate chains entwine with one-another, much as cooked spaghetti does, forming weak attractions between the chains - but with no actual bonds between the chains,
Change the double bond into a single bond and draw bonds going left and right from the carbon atoms :
Place large brackets around the structure and a subscript n and there is the repeat unit.
where R= H for ethene
= CH3 for propene
= C6H5 for phenylethene
= Cl for chloroethene
= COOCH3 for methyl acrylate
Polymer structure and properties –
When the individual alkene units join together to give a polymer they result in the formation of long chains of carbon atoms joined together. In any sample of a polymer there are many separate chains present. These chains will be of varying lengths, depending on the number of alkene units that make them up. These separate chains entwine with one-another, much as cooked spaghetti does, forming weak attractions between the chains - but with no actual bonds between the chains,