- elements, mixtures and compounds, and between metals and non-metals
Pure substances and impurities
A pure substance is a substance that has no particles of any other substance mixed with it.
An unwanted substance, mixed with a wanted substance, is called an impurity. To check if a substance is pure, you have to check its melting and boiling points.
A pure substance has a definite, sharp, melting point. When a substance is impure, the melting point falls and its boiling point rises. So the more impurity present, the wider and bigger the change in melting and boiling point.
A pure substance is a substance that has no particles of any other substance mixed with it.
An unwanted substance, mixed with a wanted substance, is called an impurity. To check if a substance is pure, you have to check its melting and boiling points.
A pure substance has a definite, sharp, melting point. When a substance is impure, the melting point falls and its boiling point rises. So the more impurity present, the wider and bigger the change in melting and boiling point.
Mixtures, Solutions, and Solvents
Mixture: Contains more the one substance. They are just mixed together and not chemically combined.
Example: Sand and water.
Solution: It is when a solute and a solvent mix. The solute dissolves in the solvent making a solution.
Example: sugar (solute) dissolves in water (solvent) making a solution of sugar and water.
The solubility of every substance is different.
To help a solute dissolve you could:
Mixture: Contains more the one substance. They are just mixed together and not chemically combined.
Example: Sand and water.
Solution: It is when a solute and a solvent mix. The solute dissolves in the solvent making a solution.
Example: sugar (solute) dissolves in water (solvent) making a solution of sugar and water.
The solubility of every substance is different.
To help a solute dissolve you could:
- Stir it
- Rise the temperature
If you add excess amount of sugar in a small amount of water...it won’t dissolve as there
is no space for it. The solution becomes saturated.
Solvent: A substance that allows solutes to dissolve in Example: Water, Ethanol
Noble Gases:
These are elements in group 8 of the periodic table. They are colorless gases. They are extremely unreactive; this is because they have their outer energy shell full with electrons. So they are stable, this is why they exist as single atoms. They have some uses however, for example argon is used in light bulbs to prevent the tungsten filament from reacting with air, making the bulb last longer. Neon is also used in the advertising and laser beams. These gases are
Helium, Neon, Argon, Krypton, Xenon and Radon (radioactive).
These are elements in group 8 of the periodic table. They are colorless gases. They are extremely unreactive; this is because they have their outer energy shell full with electrons. So they are stable, this is why they exist as single atoms. They have some uses however, for example argon is used in light bulbs to prevent the tungsten filament from reacting with air, making the bulb last longer. Neon is also used in the advertising and laser beams. These gases are
Helium, Neon, Argon, Krypton, Xenon and Radon (radioactive).
Alloys:
An alloy is a mixture of metals or metals and non-metals. Sometimes, and alloy is better than a metal because they have better properties. They are harder, more resistant to corrosion and have a more attractive appearance than the metals they are formed of.
Alloys are harder than metals because they have different sized atoms which prevent the layers from sliding over
each other.
And alloy is made by heating the metals or metals and non-metals together until they all melt, and leaving them to cool mixed.
Examples of alloys and their content:
An alloy is a mixture of metals or metals and non-metals. Sometimes, and alloy is better than a metal because they have better properties. They are harder, more resistant to corrosion and have a more attractive appearance than the metals they are formed of.
Alloys are harder than metals because they have different sized atoms which prevent the layers from sliding over
each other.
And alloy is made by heating the metals or metals and non-metals together until they all melt, and leaving them to cool mixed.
Examples of alloys and their content:
- Brass: Copper-Zinc,
- Bronze: Copper-Tin,
- Steel: Iron-Carbon,
- Stainless Steel: Iron-Carbon-Chromium-Nickel.
Bonding Structures The Ion:
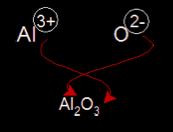
During chemical reactions involving two or more atoms, some types of atoms lose one or more electron turning into a positive ion. It is called a positive ion because when it loses electrons, the number of positive protons becomes larger than the number of negative electrons, so the overall charge of the particle is positive. If it loses two electrons its charge will be +2. If it loses 3, its charge will be +3 and so on.
Other types of atoms gain the electrons lost by the positive ions. In this case they become negative ions. This is because the number of negative electrons becomes larger than the number of positive protons, making the overall charge of the ion negative. If it gains two electrons its charge becomes -2. If it gains 3 electrons its charge becomes - 3, and so on.
During chemical reactions involving two or more atoms, some types of atoms lose one or more electron turning into a positive ion. It is called a positive ion because when it loses electrons, the number of positive protons becomes larger than the number of negative electrons, so the overall charge of the particle is positive. If it loses two electrons its charge will be +2. If it loses 3, its charge will be +3 and so on.
Other types of atoms gain the electrons lost by the positive ions. In this case they become negative ions. This is because the number of negative electrons becomes larger than the number of positive protons, making the overall charge of the ion negative. If it gains two electrons its charge becomes -2. If it gains 3 electrons its charge becomes - 3, and so on.
Ionic Bonding:
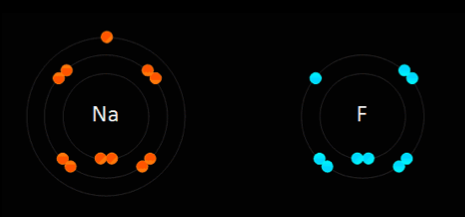
This type of bonding is based on the electrostatic force of attraction between the ions in the molecule. For example, when sodium, which is in group one and has one electron in its outer most shell, reacts with chlorine, which is in group 7 and has 7 electrons in its outer most shell, the sodium gets rid of the only electron in its outer shell, thus the sodium atom will have its second most outer shell which is full become its most outer shell forming a positive ion. The electron which is lost by the sodium atom is gained by the chlorine atom to 8 electrons, thus filling its outer most shell and becoming a negative ion. This electron transfer causes the electrostatic force of attraction which holds the oppositely charged ions together in a molecule. When an atom becomes an ion, it gets the properties of the noble gas which is nearest to it in the periodic table. Ionic bonds are only formed between metals and non-metals.
This type of bonding is based on the electrostatic force of attraction between the ions in the molecule. For example, when sodium, which is in group one and has one electron in its outer most shell, reacts with chlorine, which is in group 7 and has 7 electrons in its outer most shell, the sodium gets rid of the only electron in its outer shell, thus the sodium atom will have its second most outer shell which is full become its most outer shell forming a positive ion. The electron which is lost by the sodium atom is gained by the chlorine atom to 8 electrons, thus filling its outer most shell and becoming a negative ion. This electron transfer causes the electrostatic force of attraction which holds the oppositely charged ions together in a molecule. When an atom becomes an ion, it gets the properties of the noble gas which is nearest to it in the periodic table. Ionic bonds are only formed between metals and non-metals.
Formulae of Ionic Compounds:
To find the formula for an ionic compound, we use a method similar to cross multiplication. We multiply the valency (valency is the number of electrons an atom loses or gains to form an ion) to the other atom as shown below:
To find the formula for an ionic compound, we use a method similar to cross multiplication. We multiply the valency (valency is the number of electrons an atom loses or gains to form an ion) to the other atom as shown below:
Important notes:
- Hydrogen and the metals form positive ions
- Non-metals form negative ions, and their names end in -ide
- Group 4 and 5 do not usually form ions because they would have to lose or gain
several electrons and that takes too much energy
- Group 0 elements do not form ions; they already have full outer shells
- Some of the transition metals form more than one ion.
- Some ions can be formed from groups of joined atoms. These are called
compound ions.
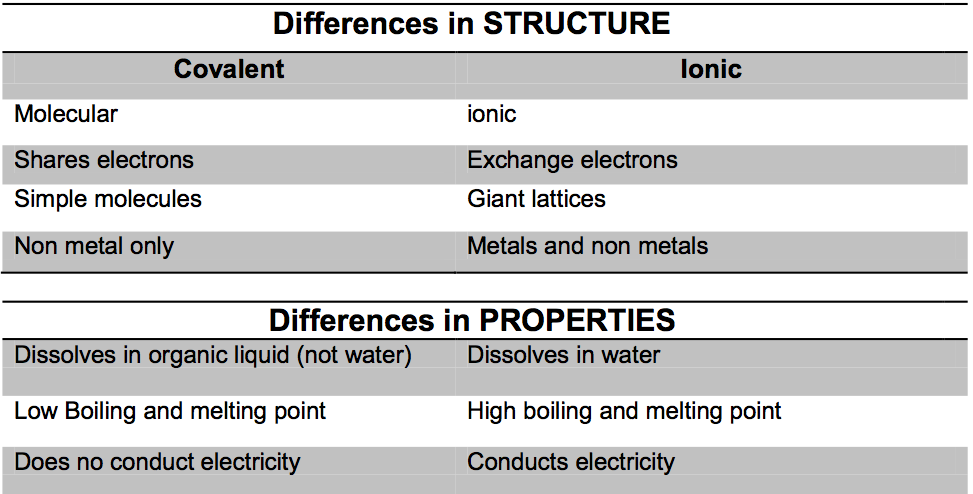
Covalent Bonding:
This type of bonding occurs between non-metals only. In order to obtain a full outer most energy shell, the atoms tend to share the electrons of their outer most energy shell, some or all of them.
Properties of covalent bonding
This is because molecules are not charged, so they cannot conduct, even when melted
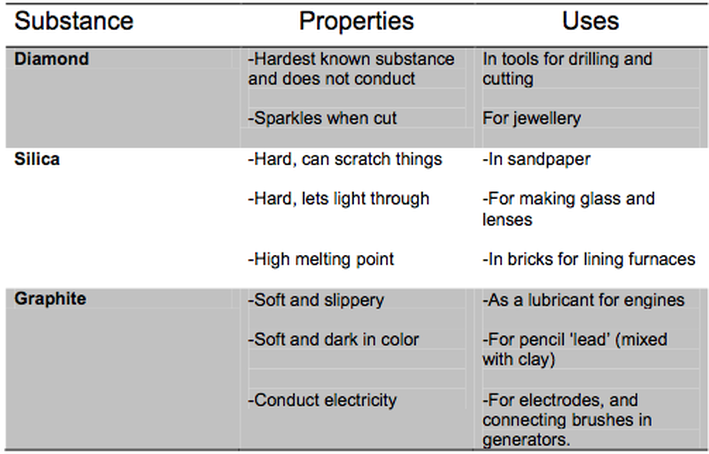
Giant covalent structures
A giant covalent structure, or macromolecules are made of billions of atoms bonded together in a covalent stricture
Molecular substances
Most molecular substances are gases or liquids at room temperature. Molecular solids are held in a lattice but the forces between the molecules are weak. All molecular solids have similar structure. The molecules are held in regular pattern in a lattice. So the solids are crystalline.
When you cool down a molecular liquid or gas the molecules lose energy so they start moving slowly and at the freezing point, they form a lattice (a good example would be ice)
This type of bonding occurs between non-metals only. In order to obtain a full outer most energy shell, the atoms tend to share the electrons of their outer most energy shell, some or all of them.
Properties of covalent bonding
- Covalent compounds have low melting and boiling point
This is because the forces between the molecules are weak.
This is because molecules are not charged, so they cannot conduct, even when melted
Giant covalent structures
A giant covalent structure, or macromolecules are made of billions of atoms bonded together in a covalent stricture
Molecular substances
Most molecular substances are gases or liquids at room temperature. Molecular solids are held in a lattice but the forces between the molecules are weak. All molecular solids have similar structure. The molecules are held in regular pattern in a lattice. So the solids are crystalline.
When you cool down a molecular liquid or gas the molecules lose energy so they start moving slowly and at the freezing point, they form a lattice (a good example would be ice)
Comparing Bonds
Metallic bonding
Metals form giant structures in which electrons in the outer shells of the metal atoms are free to move. The metallic bond is the force of attraction between these free electrons and metal ions. Metallic bonds are strong, so metals can maintain a regular structure and usually have high melting and boiling points.
Metals form giant structures in which electrons in the outer shells of the metal atoms are free to move. The metallic bond is the force of attraction between these free electrons and metal ions. Metallic bonds are strong, so metals can maintain a regular structure and usually have high melting and boiling points.