Alcohol
Alcohols all have an -OH group and their names follow this pattern, CH3OH - methanol
C2H5OH - ethanol C3H7OH - propanol C4H9OH - butanol C5H11OH - pentanol
The general chemical formula for an alcohol is CnH2nOH.
Preparation of ethanol by fermentation –
Ethanol is prepared in the laboratory and in the alcoholic drinks industry, by the process of fermentation. This involves the use of an enzyme ( yeast ) that changes a carbohydrate, e.g. sucrose, into ethanol and carbon dioxide gas,
C6H12O6(aq) 2CH3CH2OH(aq) + 2CO2(g)
The yeast used requires a certain temperature to be active - somewhere between 15 and 37 °C. Too high a temperature and the yeast "dies" and too low a temperature causes the yeast to become dormant.
The production of carbon dioxide gas can be monitored by bubbling any gases produced during the reaction through limewater (calcium hydroxide(aq) ). The formation of a white precipitate (calcium carbonate ) in the limewater shows that carbon dioxide has been given off.
Dehydration of ethanol -
Experimental sheet for the dehydration of ethanol.
All alcohols contain hydrogen and oxygen ( as well as carbon ) and these atoms can be removed from an alcohol as a molecule of water ( H2O ). This type of reaction is called dehydration. It can be accomplished by passing alcohol vapour over a heated aluminium oxide catalyst.
e.g. ethanol can be turned into ethene,
CH3CH2OH(g) CH2=CH2(g) + H2O(g)
Oxidation of ethanol -
Experimental sheet for the oxidation of ethanol.
Oxidation can be defined as the addition of oxygen to a substance. This can be accomplished with alcohols by the use of acidified potassium dichromate(VI)(aq). This turns the alcohol into a carboxylic acid.
e.g. ethanol can be turned into ethanoic acid,
C2H5OH - ethanol C3H7OH - propanol C4H9OH - butanol C5H11OH - pentanol
The general chemical formula for an alcohol is CnH2nOH.
Preparation of ethanol by fermentation –
Ethanol is prepared in the laboratory and in the alcoholic drinks industry, by the process of fermentation. This involves the use of an enzyme ( yeast ) that changes a carbohydrate, e.g. sucrose, into ethanol and carbon dioxide gas,
C6H12O6(aq) 2CH3CH2OH(aq) + 2CO2(g)
The yeast used requires a certain temperature to be active - somewhere between 15 and 37 °C. Too high a temperature and the yeast "dies" and too low a temperature causes the yeast to become dormant.
The production of carbon dioxide gas can be monitored by bubbling any gases produced during the reaction through limewater (calcium hydroxide(aq) ). The formation of a white precipitate (calcium carbonate ) in the limewater shows that carbon dioxide has been given off.
Dehydration of ethanol -
Experimental sheet for the dehydration of ethanol.
All alcohols contain hydrogen and oxygen ( as well as carbon ) and these atoms can be removed from an alcohol as a molecule of water ( H2O ). This type of reaction is called dehydration. It can be accomplished by passing alcohol vapour over a heated aluminium oxide catalyst.
e.g. ethanol can be turned into ethene,
CH3CH2OH(g) CH2=CH2(g) + H2O(g)
Oxidation of ethanol -
Experimental sheet for the oxidation of ethanol.
Oxidation can be defined as the addition of oxygen to a substance. This can be accomplished with alcohols by the use of acidified potassium dichromate(VI)(aq). This turns the alcohol into a carboxylic acid.
e.g. ethanol can be turned into ethanoic acid,
Carboxylic Acids
Carboxylic acids all have the -COOH structural group in them and their names follow this pattern, HCOOH - methanoic acid
CH3COOH - ethanoic acid
C2H5COOH - propanoic acid
C3H7COOH - butanoic acid
C4H9COOH - pentanoic acid
The general chemical formula for a carboxylic acid is CnH2n1-COOH.
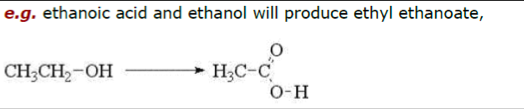
The Esterification of Ethanol to Ethyl Ethanoate
Introduction
Apart from the normal reactions of acids, such as reactions with metals and bases, ethanoic acid (along with all the carboxylic acids) will react with ethanol (or other alcohols) to give ethyl ethanoate (an ester) in a simple process.
Carboxylic acids all have the -COOH structural group in them and their names follow this pattern, HCOOH - methanoic acid
CH3COOH - ethanoic acid
C2H5COOH - propanoic acid
C3H7COOH - butanoic acid
C4H9COOH - pentanoic acid
The general chemical formula for a carboxylic acid is CnH2n1-COOH.
The Esterification of Ethanol to Ethyl Ethanoate
Introduction
Apart from the normal reactions of acids, such as reactions with metals and bases, ethanoic acid (along with all the carboxylic acids) will react with ethanol (or other alcohols) to give ethyl ethanoate (an ester) in a simple process.
Condensation polymerisation :
As well as the addition polymers formed from alkenes and free radical initiators already mentioned, there is another method of preparing long chain polymers.
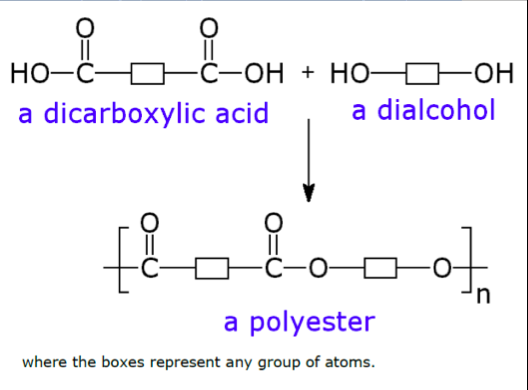
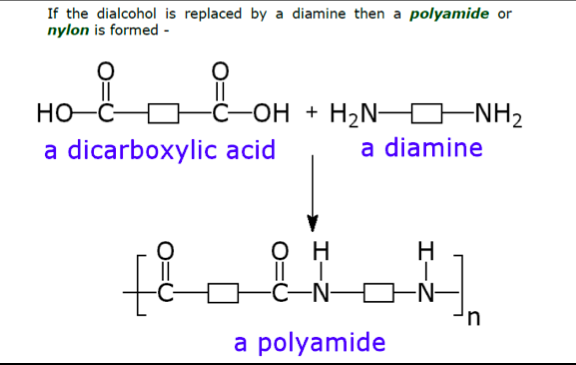
This second method of polymerisation relies on the reaction between a dicarboxylic acid and an dialcohol ( or a diamine ) and is called condensation polymerisation since water is released during the formation of the polymer chains.
A monocarboxylic acid will react with an alcohol to give an ester ( see equation above).
If a molecule had two carboxylic acid groups on it, one at each end, and it reacted with a molecule with two -OH groups on it then many ester groups, i.e. a polyester, would be formed and long chains produced –
As well as the addition polymers formed from alkenes and free radical initiators already mentioned, there is another method of preparing long chain polymers.
This second method of polymerisation relies on the reaction between a dicarboxylic acid and an dialcohol ( or a diamine ) and is called condensation polymerisation since water is released during the formation of the polymer chains.
A monocarboxylic acid will react with an alcohol to give an ester ( see equation above).
If a molecule had two carboxylic acid groups on it, one at each end, and it reacted with a molecule with two -OH groups on it then many ester groups, i.e. a polyester, would be formed and long chains produced –
Natural condensation polymers :
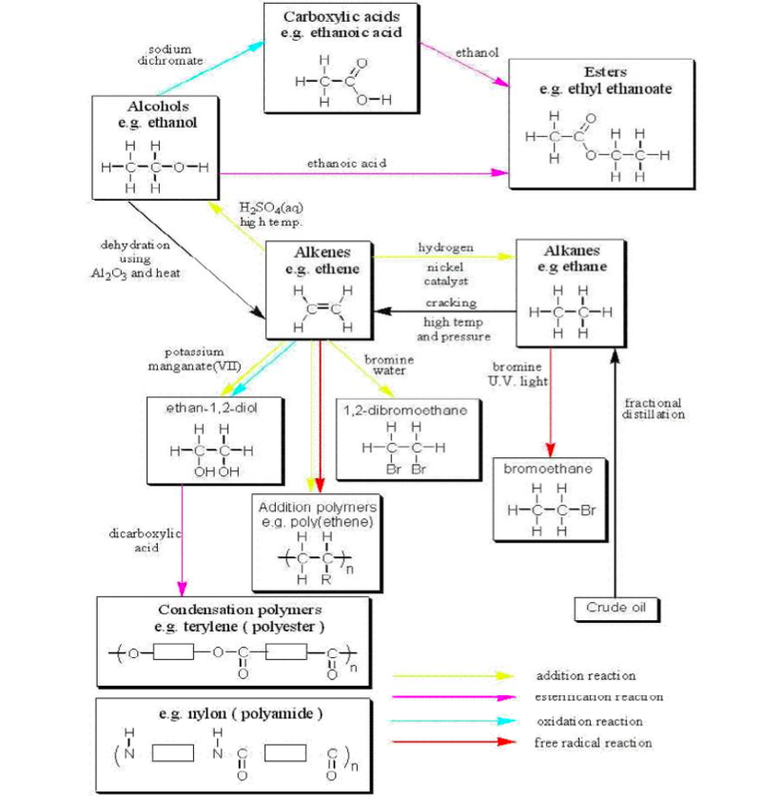
The above picture encompasses only the synthetic part of the organic work. There are a number of natural polymers required.
These are :
(i) Fats :
These natural materials contain the ester link found in the synthetic polyesters shown above.
They may be hydrolysed ( broken down ) by a reaction with sodium hydroxide (a strong base) and heat. Once hydrolysed they form soaps ( sodium salts of carboxylic acids ) and glycerol ( propan-1,2,3-triol ).
Proteins :
These naturally occurring materials contain the amide link found in the synthetic polyamides shown above.
These compounds may also be hydrolysed by a reaction with enzymes and/or aqueous acid. Proteins in the food we ingest are broken down by stomach acids and enzymes which work at body temperature.
Once hydrolysed they form amino acids which can then be used by the human body to prepare vital chemicals needed to sustain life.
Saponification :
Saponification means "soap-making" and is a reaction in which a fat, or oil, is turned into a salt of a carboxylic acid. The oil is heated with a concentrated solution of a caustic base, such as sodium hydroxide. The base breaks down the ester links, forming alcohol groups and carboxylate ion groups on different molecules.
The above picture encompasses only the synthetic part of the organic work. There are a number of natural polymers required.
These are :
(i) Fats :
These natural materials contain the ester link found in the synthetic polyesters shown above.
They may be hydrolysed ( broken down ) by a reaction with sodium hydroxide (a strong base) and heat. Once hydrolysed they form soaps ( sodium salts of carboxylic acids ) and glycerol ( propan-1,2,3-triol ).
Proteins :
These naturally occurring materials contain the amide link found in the synthetic polyamides shown above.
These compounds may also be hydrolysed by a reaction with enzymes and/or aqueous acid. Proteins in the food we ingest are broken down by stomach acids and enzymes which work at body temperature.
Once hydrolysed they form amino acids which can then be used by the human body to prepare vital chemicals needed to sustain life.
Saponification :
Saponification means "soap-making" and is a reaction in which a fat, or oil, is turned into a salt of a carboxylic acid. The oil is heated with a concentrated solution of a caustic base, such as sodium hydroxide. The base breaks down the ester links, forming alcohol groups and carboxylate ion groups on different molecules.