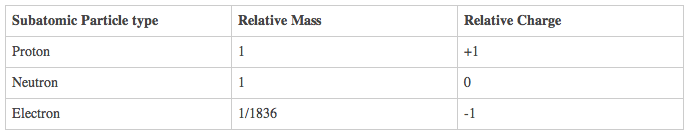3.3. Atomic Structure and the Periodic Table1 Describe the structure of an atom in terms of electrons and a nucleus containing protons and neutrons.
An atom consists of what we call, Subatomic Particles, the things that make up the atoms.
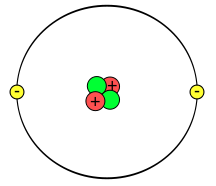
Look at the figure above. Here is the basic model of an atom, and it consists of three different sub-atomic particles. The green circle is called the Neutron. The Red circle is called a Proton. And lastly, the yellow circle is called a Electron.
So, lets examine this qualitatively.
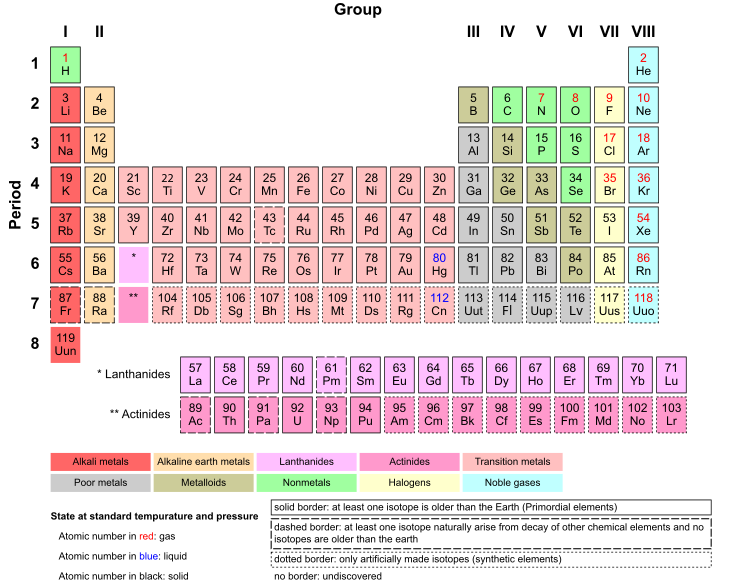
To do this, we need a copy of a periodic table.
So, lets examine this qualitatively.
- The small circles within the large circle is located in a region called the Nucleus. The nucleus is home to the protons and neutrons.
- Orbiting the nucleus are the electrons. These are called “Electron Orbitals”
To do this, we need a copy of a periodic table.
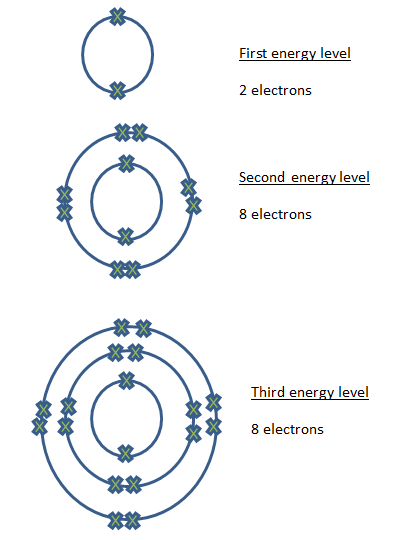
Electrons are arranged in shells. Each group is equivalent to one energy level and when you reach the end of the period of the group, you start off at a new group.
- 1st energy level holds up to 2 electrons
- 2nd energy level holds up to 8 electrons
- 3rd energy level holds up to 8 electrons.
f an atom is in its 3rd energy level holding 8 electrons, the addition of 1 more electrons will push it to the 4th energy level.
The atoms at the end of the periodic table have a full electron shell, so basically, no more additional electrons may be added on and it has no valence electrons. We call these Noble Gases.
3 State the relative charges and approximate relative masses of protons, neutrons and
electrons.
The atoms at the end of the periodic table have a full electron shell, so basically, no more additional electrons may be added on and it has no valence electrons. We call these Noble Gases.
3 State the relative charges and approximate relative masses of protons, neutrons and
electrons.
4 Define proton number and nucleon number.
- Nucleon number (A): The number of protons and neutrons in a nucleus
- Proton number (Z): The number of protons in the nucleus.
- Neutron number (N) : The number of neutrons in the nucleus.
5 Use proton number and the simple structure of atoms to explain the basis of the Periodic Table (see C9), with special reference to the elements of proton number 1 to 20.
Each time you go across a period, lets say Helium, the atom has one more proton that the element on the left. For Example, Hydrogen has one proton, and Helium has two. Additionally, Lithium is now down a group so it has 3 protons. Moving along the period, you have Beryllium, Boron, Carbon, Nitrogen, Oxygen and so on. After we reach Ne (Neon) which has 10 electrons, we move on to the next group and a different energy level of electrons.
Each time you go across a period, lets say Helium, the atom has one more proton that the element on the left. For Example, Hydrogen has one proton, and Helium has two. Additionally, Lithium is now down a group so it has 3 protons. Moving along the period, you have Beryllium, Boron, Carbon, Nitrogen, Oxygen and so on. After we reach Ne (Neon) which has 10 electrons, we move on to the next group and a different energy level of electrons.