Chapter 10: Metals
The Reactivity Series of Metals:
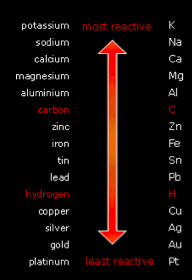
The reactivity series is metal is an arrangement of the metals (and carbon and hydrogen) in order of their reactivity starting with the most reactive metal at the top and ending with the least reactive metal at the bottom.
The reactivity of a metal is determined by its ability to form a positive ion. For example, potassium is extremely reactive because it has only one valence electron, so it is very easy to lose it forming a positive ion.
One the other hand, copper is a weakly reactive metal because it has more valence electrons so it is harder for it to become a positive ion.
Reactions of Metals:
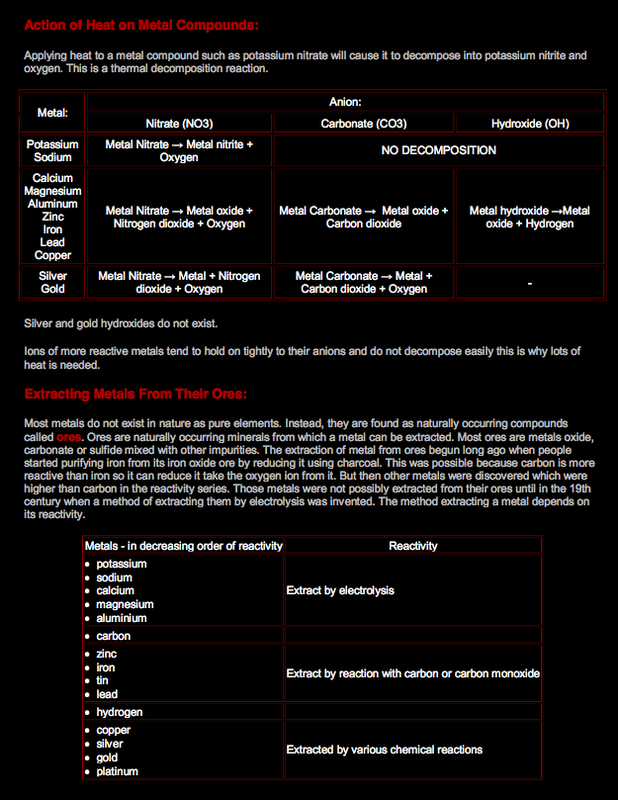
The reactivity series of metals was deduced by performing several experiments in the lab which enabled scientists to arrange metals according to their reactivity with dilute acid, oxygen (air), and water.
The reactivity series is metal is an arrangement of the metals (and carbon and hydrogen) in order of their reactivity starting with the most reactive metal at the top and ending with the least reactive metal at the bottom.
The reactivity of a metal is determined by its ability to form a positive ion. For example, potassium is extremely reactive because it has only one valence electron, so it is very easy to lose it forming a positive ion.
One the other hand, copper is a weakly reactive metal because it has more valence electrons so it is harder for it to become a positive ion.
Reactions of Metals:
The reactivity series of metals was deduced by performing several experiments in the lab which enabled scientists to arrange metals according to their reactivity with dilute acid, oxygen (air), and water.
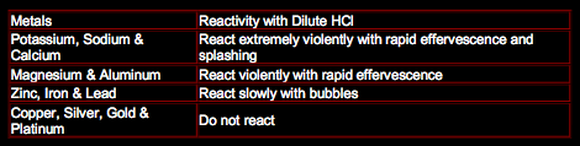
Reactions with Dilute Hydrochloric Acid:
The photo on the right shows magnesium reacting with dilute hydrochloric acid. Those effervescences are caused by the evolution of hydrogen gas, which is a product in this reaction. This reaction was repeated using the other metals of the reactivity series. The rate of evolution of hydrogen gas in each experiment was measured. The metals were arranged in order of reactivity starting with the most reactive metal which had the highest rate of effervescence of hydrogen gas. The rate of effervescence is also the rate of this reaction is measured by measuring the volume of hydrogen produced per unit time.
The photo on the right shows magnesium reacting with dilute hydrochloric acid. Those effervescences are caused by the evolution of hydrogen gas, which is a product in this reaction. This reaction was repeated using the other metals of the reactivity series. The rate of evolution of hydrogen gas in each experiment was measured. The metals were arranged in order of reactivity starting with the most reactive metal which had the highest rate of effervescence of hydrogen gas. The rate of effervescence is also the rate of this reaction is measured by measuring the volume of hydrogen produced per unit time.
Reactions with Oxygen in Air:
Most metals react with oxygen from air forming a metal oxide. You have previously studied that metal oxides are basic oxides and that some of them are insoluble in water and some of them are soluble in water forming an alkaline
solution. The most reactive metals like potassium, sodium, calcium and magnesium react with oxygen with a very bright flame and producing white ashes and their oxides are soluble. Moderately reactive metals like aluminum and zinc react with oxygen forming white powdered ashes but their oxides are insoluble. Iron and copper react very slowly with oxygen. The result of iron oxygen reactions is rust which is reddish brown iron oxide. When a copper lump reacts with oxygen, a white layer of black copper oxide forms on it. When the lump gets covered by this layer;
Most metals react with oxygen from air forming a metal oxide. You have previously studied that metal oxides are basic oxides and that some of them are insoluble in water and some of them are soluble in water forming an alkaline
solution. The most reactive metals like potassium, sodium, calcium and magnesium react with oxygen with a very bright flame and producing white ashes and their oxides are soluble. Moderately reactive metals like aluminum and zinc react with oxygen forming white powdered ashes but their oxides are insoluble. Iron and copper react very slowly with oxygen. The result of iron oxygen reactions is rust which is reddish brown iron oxide. When a copper lump reacts with oxygen, a white layer of black copper oxide forms on it. When the lump gets covered by this layer;
Competition Reactions in Solid State:
Previously you’ve studied displacement reactions which are pre-formed in aqueous states. A very similar reaction takes place in the solid state, it is called thermite reaction. This reaction is used to repair damaged railway lines. In this reaction, aluminum and iron (III) oxide are the reactants. In the reaction, aluminum removes the oxygen ion from iron and bonds with it. This happens because aluminum is more reactive than iron. The products are aluminum oxide and iron in molten form. In the fixing procedure, the reactants are put in the cut in the railway line and the reaction is triggered by heating using a magnesium fuse. The reaction leaves aluminum oxide and molten iron with then condenses in the cut welding it. Like displacement reactions, this reaction is exothermic.
2Al + Fe2O3 → Al2O3 +2Fe Competition Reactions in Aqueous State:
These are ordinary displacement reactions in which the two positive ions compete for the negative ion. The ion of the more reactive metal wins. Zinc is higher than copper in the reactivity series. If zinc is added to a solution of copper nitrate, a displacement reaction will take place in which the zinc will displace the copper ion from the solution in its salt. The products of this reaction are zinc nitrate and copper. Copper salt solutions have a blue color which fades away as the reaction proceeds because the concentration of the copper salt decreases. This type of reaction also
helped in confirming reactivity of metals since the more reactive metal displaces the less reactive one.
Zn + Cu(NO3)2 → Zn(NO3)2 + Cu
Previously you’ve studied displacement reactions which are pre-formed in aqueous states. A very similar reaction takes place in the solid state, it is called thermite reaction. This reaction is used to repair damaged railway lines. In this reaction, aluminum and iron (III) oxide are the reactants. In the reaction, aluminum removes the oxygen ion from iron and bonds with it. This happens because aluminum is more reactive than iron. The products are aluminum oxide and iron in molten form. In the fixing procedure, the reactants are put in the cut in the railway line and the reaction is triggered by heating using a magnesium fuse. The reaction leaves aluminum oxide and molten iron with then condenses in the cut welding it. Like displacement reactions, this reaction is exothermic.
2Al + Fe2O3 → Al2O3 +2Fe Competition Reactions in Aqueous State:
These are ordinary displacement reactions in which the two positive ions compete for the negative ion. The ion of the more reactive metal wins. Zinc is higher than copper in the reactivity series. If zinc is added to a solution of copper nitrate, a displacement reaction will take place in which the zinc will displace the copper ion from the solution in its salt. The products of this reaction are zinc nitrate and copper. Copper salt solutions have a blue color which fades away as the reaction proceeds because the concentration of the copper salt decreases. This type of reaction also
helped in confirming reactivity of metals since the more reactive metal displaces the less reactive one.
Zn + Cu(NO3)2 → Zn(NO3)2 + Cu
Extraction of Aluminium:
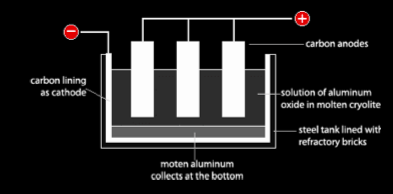
Aluminum exists naturally as aluminum oxide (alumina) in its ore, which is called bauxite. Because aluminum is a very reactive metal, it holds on very tightly to the anion it bonds with, which is oxide in this case. This is why the best way to extract and purify aluminum is by electrolysis in a cell like the one below.
Aluminum exists naturally as aluminum oxide (alumina) in its ore, which is called bauxite. Because aluminum is a very reactive metal, it holds on very tightly to the anion it bonds with, which is oxide in this case. This is why the best way to extract and purify aluminum is by electrolysis in a cell like the one below.
In this cell, the electrodes are made of graphite (Carbon). The cathode is a layer at the bottom of the cell and the anodes are bars dipped in the electrolyte. The electrolyte in this process is a molten mixture of aluminum oxide and
cryolite. Aluminum oxide by its self has a very high melting point of 2050oC which is higher than the melting point of the steel container in which this process is done. That means the steel container will melt before the aluminum oxide. This is why aluminum oxide is mixed with cryolite which decreases the melting point of it to under 1000oC, thus saving a lot of money because heating is expensive and preventing the steel container from melting. Heat must be continuously supplied to the mixture to keep it molten. Aluminum oxide does not conduct electricity when solid because it does not have free mobile ions to carry the charge.
Oxygen gas which evolves reacts with carbon from the cathode forming CO2. The cathode gets worn away. To solve this, the cathode is replaced at regular intervals. Heat supply is very expensive; this is why cryolite is used to decrease the melting point of aluminum oxide and this process is done in plants which use hydroelectric energy
because it is cheap.
Uses of aluminium:
Strengthened with steel core
The ore of iron is called hematite. It consists of 60% iron in form of Iron oxide (Fe2O3) with other impurities such as silicon
oxide (SiO2). This process takes place in a tower called a Blast furnace.
cryolite. Aluminum oxide by its self has a very high melting point of 2050oC which is higher than the melting point of the steel container in which this process is done. That means the steel container will melt before the aluminum oxide. This is why aluminum oxide is mixed with cryolite which decreases the melting point of it to under 1000oC, thus saving a lot of money because heating is expensive and preventing the steel container from melting. Heat must be continuously supplied to the mixture to keep it molten. Aluminum oxide does not conduct electricity when solid because it does not have free mobile ions to carry the charge.
- Aluminum oxide is purified from impurities of oxide by adding sodium hydroxide
- Aluminum oxide is mixed with cryolite and put in the electrolysis cell
- Heat is given in until the mixture becomes molten
- Electrolysis start
- Oxide ions get attracted to the anode and discharged (oxidation); 2O2-, 4e → O2
- Aluminum ions get attracted to the cathode and discharged and settle at the bottom
of the container (reduction); Al3+ + 3e → Al
- Oxygen gas evolves and is collected with waste gases
- Aluminum is sucked out of the container at regular intervals
Oxygen gas which evolves reacts with carbon from the cathode forming CO2. The cathode gets worn away. To solve this, the cathode is replaced at regular intervals. Heat supply is very expensive; this is why cryolite is used to decrease the melting point of aluminum oxide and this process is done in plants which use hydroelectric energy
because it is cheap.
Uses of aluminium:
- Construction of air-craft bodies because aluminum is very strong and very light and it is resistant to corrosion
- Food containers because it is resistant to corrosion
Strengthened with steel core
The ore of iron is called hematite. It consists of 60% iron in form of Iron oxide (Fe2O3) with other impurities such as silicon
oxide (SiO2). This process takes place in a tower called a Blast furnace.
- Substances are put in the blast furnace
- The process starts by blowing in hot air at the bottom of the furnace
- Coke burns in oxygen from the hot air producing carbon dioxide; C + O2 → CO2
- Heat makes lime stone decompose into calcium oxide and carbon dioxide; CaCO3 → CaO + CO2
- Carbon dioxide produced goes up the furnace and reacts with more coke up there producing
carbon monoxide; CO2 + C → 2CO
- Carbon monoxide is a reducing agent. It rises further up the furnace where it meets iron oxide and starts
reducing it producing iron and carbon dioxide; Fe2O3 + 3CO → 2Fe + 3CO2
- Calcium oxide which was produced from the thermal decomposition of lime stone is a base. It reacts with
impurities of hematite such as silicon oxide which is acidic forming calcium silicate which is called slag; CaO
+ SiO2 → CaSiO3
- Molten Iron and slag produced trickles down and settles at the bottom of the furnace. Iron is denser than slag
so it settles beneath it.
Iron and slag are tapped off separately at regular intervals and pure iron is collected alone
- Waste gases such as carbon dioxide formed in the process and nitrogen and other gases from air blown in
escape at the top of the furnace.