- Describe methods of purification by the use of a suitable solvent, filtration, crystallisation, distillation
A pure substance is a substance that has no particles of any other substance mixed with it.
An unwanted substance, mixed with a wanted substance, is called an impurity. To check if a substance is pure, you have to check its melting and boiling points.
A pure substance has a definite, sharp, melting point. When a substance is impure, the melting point falls and its boiling point rises. So the more impurity present, the wider and bigger the change in melting and boiling point.
An unwanted substance, mixed with a wanted substance, is called an impurity. To check if a substance is pure, you have to check its melting and boiling points.
A pure substance has a definite, sharp, melting point. When a substance is impure, the melting point falls and its boiling point rises. So the more impurity present, the wider and bigger the change in melting and boiling point.
Separation methods:
Filter ------------------------- Solid from liquid
Centrifuge ------------------ Solid from liquid
Evaporation ---------------- Solid from its solution
Crystallisation -------------- Solid from its solution
Distillation ------------------ Solvent from a solution
Fractional distillation ----- Liquid from each other
Chromatography ---------- Different substances from a solution
Centrifuge ------------------ Solid from liquid
Evaporation ---------------- Solid from its solution
Crystallisation -------------- Solid from its solution
Distillation ------------------ Solvent from a solution
Fractional distillation ----- Liquid from each other
Chromatography ---------- Different substances from a solution
1. Filtering
Example:
A mixture of chalk and water...
1. A filter paper is placed in a funnel, the funnel placed on a flask. 2. The mixture is poured on the filter paper.
The chalk (the residue) will remain in the filter paper and the water (the filtrate) will fall down in the flask.
Example:
A mixture of chalk and water...
1. A filter paper is placed in a funnel, the funnel placed on a flask. 2. The mixture is poured on the filter paper.
The chalk (the residue) will remain in the filter paper and the water (the filtrate) will fall down in the flask.
2. Centrifuging
This method is used to separate small amounts of solid and liquid. Inside a centrifuge (it’s a machine), test tubes are spun very fast so the solid gets flung to the bottom.
This method is used to separate small amounts of solid and liquid. Inside a centrifuge (it’s a machine), test tubes are spun very fast so the solid gets flung to the bottom.
3. Evaporation
This method is used to separate a solution in which the solid is dissolved in the liquid. 1. The solution is heated so that the liquid evaporates and the solid remains in the
bottom of the evaporating dish.
This method is used to separate a solution in which the solid is dissolved in the liquid. 1. The solution is heated so that the liquid evaporates and the solid remains in the
bottom of the evaporating dish.
4. Crystallization
This method is similar to evaporation but here the solid forms crystals then the crystals are left to dry.
Separating a mixture of two solids
This method is similar to evaporation but here the solid forms crystals then the crystals are left to dry.
Separating a mixture of two solids
- This can be done by dissolving one in an appropriate solvent.
- Then filtering one and extracting the other from the solution by evaporation.
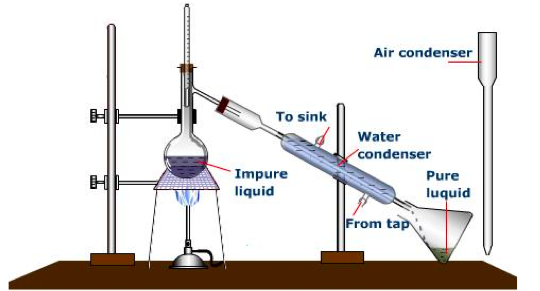
5. Simple distillation
- The impure liquid is heated.
- It boils, and steam rises into the condenser.
- The impurities are left behind.
- The condenser is cold so the steam condenses to the pure liquid and it drops out
on the beaker.
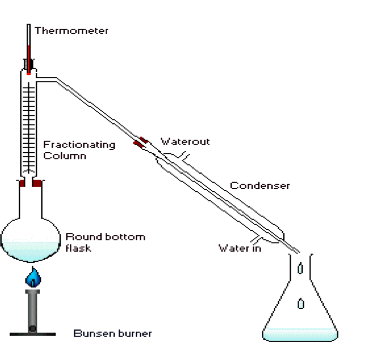
6. Fractional distillation
- The mixture is heated.
- The wanted substance boils and evaporates
(some of the unwanted liquid will evaporate
too) and rises up the column.
- The substance will condense on the beads
in the column causing them to heat.
- When the beads reach a certain temperature
when the wanted liquid wont condense anymore (That’s the boiling point) it will rise while the unwanted liquid will condense and drop. The wanted liquid will make its way through the condenser where it will condense and drop down in the beaker.
7. Chromatography
This method is used to separate a mixture of substances. For example you can use it to find how many coloured substances there are in black ink.
Steps:
1. Drop the black ink on to the center of a filter paper and allow it to dry.
2. Drop water on to the ink spot, one drop at a time.
3. Suppose there are three rings: yellow, red and blue. This shows the ink contains 3 coloured substances.
The substances travel across the paper at different rates. That’s why they separate into rings. The filter paper showing the separate substances is called a chromatogram. This method works because different substances travel at different speeds because they have different levels of attraction to it.
Uses of chromatography:
This method is used to separate a mixture of substances. For example you can use it to find how many coloured substances there are in black ink.
Steps:
1. Drop the black ink on to the center of a filter paper and allow it to dry.
2. Drop water on to the ink spot, one drop at a time.
3. Suppose there are three rings: yellow, red and blue. This shows the ink contains 3 coloured substances.
The substances travel across the paper at different rates. That’s why they separate into rings. The filter paper showing the separate substances is called a chromatogram. This method works because different substances travel at different speeds because they have different levels of attraction to it.
Uses of chromatography:
- Separate mixtures of substances
- Purify a substance by separating the impurities from it
- Identify a substance