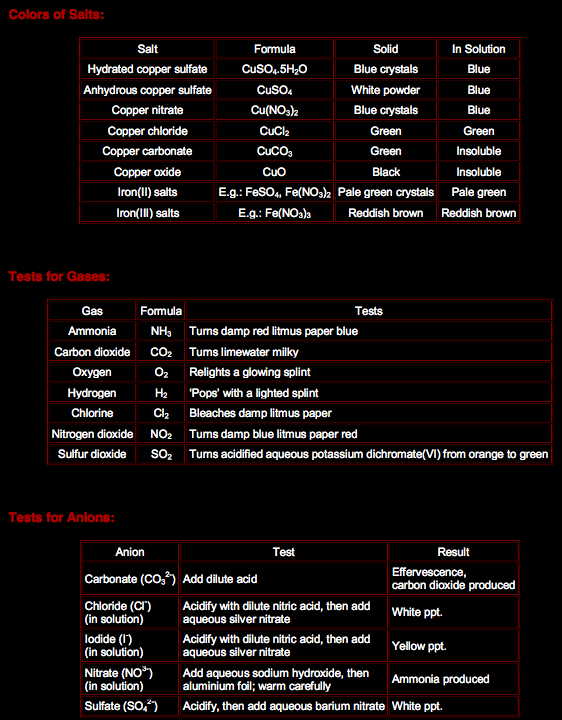
Titration Method:
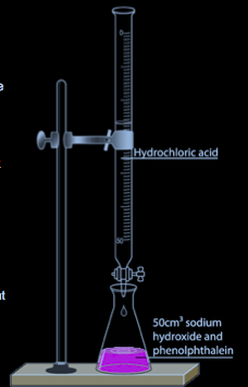
This is a method to make a neutralization reaction between a base and an acid producing a salt without any excess. In this method, the experiment is preformed twice, the first time is to find the amounts of reactants to use, and the second experiment is the actual one.
1st Experiment:
1st Experiment:
- Add 50 cm3 of sodium hydroxide using a pipette to be accurate to flask
- Add 5 drops of phenolphthalein indicator to the sodium hydroxide. The
solution turns pink indicating presence of a base
- Fill a burette to zero mark with hydrochloric acid
- Add drops of the acid to conical flask
- The pink color of the solution becomes lighter
- When the solution turns colorless, stop adding the acid (End point: is
the point at which every base molecule is neutralized by an acid
molecule)
- Record the amount of hydrochloric acid used and repeat the experiment
without using the indicator
- After the 2nd experiment, you will have a sodium chloride solution.
Evaporate it till dryness to obtain powdered sodium chloride or crystalize it to obtain sodium chloride crystals
- Preparing Insoluble Salts:
Precipitation Method:
A precipitation reaction is a reaction between two soluble salts. The products of a precipitation reaction are two other salts, one of them is soluble and one is insoluble (precipitate).
Example: To obtain barium sulfate salt given barium chloride and sodium sulfate:
BaCl2 + Na2SO4 → BaSO4 + 2NaCl Ionic Equation: Ba2+ + SO42- → BaSO4
- Add the two salt solutions in a beaker
- When the reaction is over, filter and take the residue
- Wash the residue with distilled water and dry it in the oven
Observations:
You know the reaction is over when:
- Temperature increases
- An insoluble solid precipitate (Barium sulfate) forms
- The temperature stops rising
- No more precipitate is being formed
f the pH of the soil goes below or above 7, it has to be neutralized using an acid or a base. If the pH of the soil goes below 7, calcium carbonate (lime stone) is used to neutralize it. The pH of the soil can be measured by taking a sample from the soil, crushing it, dissolving in water then measuring the pH of the solution
The Periodic Table of Elements:
The periodic table is a table containing all elements arranged in ascending order from the one with lowest atomic number to the one with highest atomic number.
There are there are 8 vertical groups (Columns) in the periodic table. The group number is equal to the number of electrons in the outer most energy shell of the atoms of the elements in the group. This is why elements of the same
group share the same chemical properties.
There are seven horizontal periods (rows) in the periodic table. The period number is also the number of occupied energy shells in the atoms of the elements in the period.
So if an element has 3 valence electrons, it will be in group 3. And if it has 4 occupied energy shells, it will be in period 4.
We have two types of elements in the periodic table. These are Metals and Non-metals. As we move in the periodic table from the left to the right, the metallic properties of elements decrease. Metals include Magnesium, Calcium, and Sodium. Non-metals include Carbon, Oxygen and Chlorine. All metals are solid. All non-metals are either solid or gas, except for bromine which is liquid.
The periodic table is a table containing all elements arranged in ascending order from the one with lowest atomic number to the one with highest atomic number.
There are there are 8 vertical groups (Columns) in the periodic table. The group number is equal to the number of electrons in the outer most energy shell of the atoms of the elements in the group. This is why elements of the same
group share the same chemical properties.
There are seven horizontal periods (rows) in the periodic table. The period number is also the number of occupied energy shells in the atoms of the elements in the period.
So if an element has 3 valence electrons, it will be in group 3. And if it has 4 occupied energy shells, it will be in period 4.
We have two types of elements in the periodic table. These are Metals and Non-metals. As we move in the periodic table from the left to the right, the metallic properties of elements decrease. Metals include Magnesium, Calcium, and Sodium. Non-metals include Carbon, Oxygen and Chlorine. All metals are solid. All non-metals are either solid or gas, except for bromine which is liquid.